Reactions of Carbohydrates ? What is the significance of each of the components? Answer- Reducing sugars are usually detected by Benedict’s reagent,which contains copper sulphate, sodium citrate and sodium carbonate. Sodium carbonate makes the medium alkaline. Copper sulphate furnishes Cu. Reducing sugars under alkaline conditions tautomerise and form enediols. 
Enediols are powerful reducing agents. They can reduce cupric ions to cuprous form which is the basis for Benedict’s reaction. The alkaline conditions of this test also cause isomeric transformation of ketoses to aldoses, resulting in all mono- saccharides and most disaccharides to respond to this test by reducing the blue Cu. Cu. 2O), a brick red- orange precipitate. 
This solution has been used in clinical laboratories for testing urine. Q. 2- Why is Benedict’s test considered a semi quantitative test? Answer- In Benedict’s test, when fixed volume of urine (8 drops) and Benedict’s reagent(5 ml) are boiled for 2 minutes, different colored precipitated are obtained. 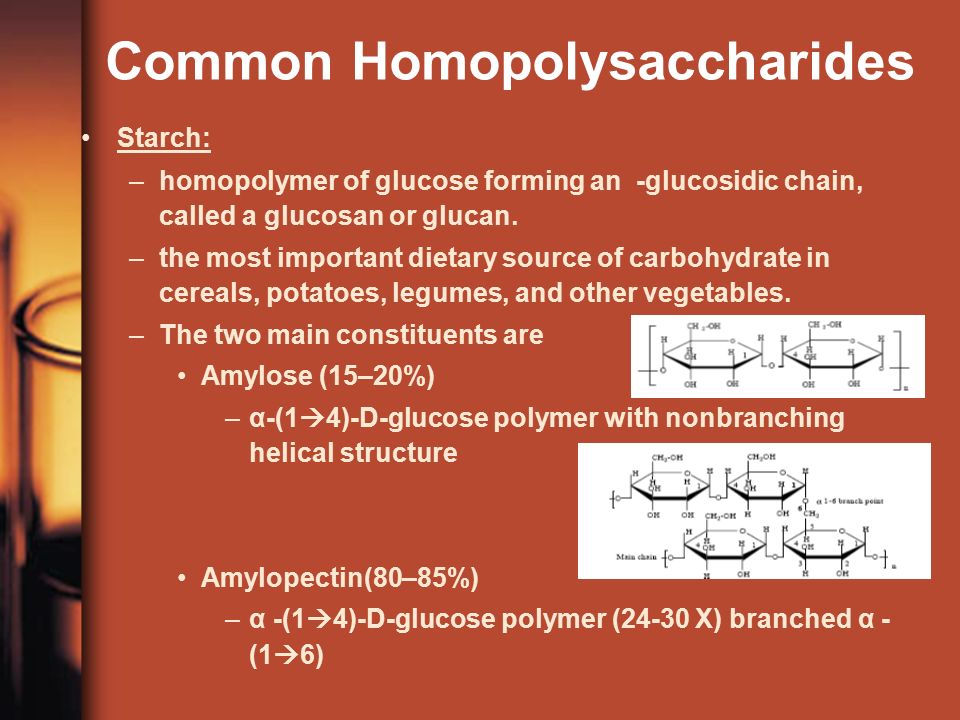
274 Asia Pac J Clin Nutr 2010;19 (2):274-282 Short Communication Sources and intake of resistant starch in the Chinese diet Liyong Chen MD 1, Ruiping Liu MD2. In this lesson, you'll learn what starch is composed of and some foods that contain it. In addition you will discover more about the chemical. Study Campbell Biology: Chapter 5 Test Preparation flashcards taken from chapter 5 of the book Campbell Biology. Hello im struggling with eczema ive been on the aip diet for about month. Ive got still problems with my digestive system like gases and gurgling. Oxford Cambridge and RSA Examinations. Advanced Subsidiary GCE. F212: Molecules, Biodiversity, Food and Health. Mark Scheme for January 2011.The precipitate color can vary between green, yellow, orange or red. The color of the precipitate signifies the rough estimate of the amount of reducing sugar present in the given sample. The interpretations are made as follows- Color of the precipitate Amount of reducing sugar present in the given sample. 
Green color of solution Up to 0. Green precipitate 0. Yellow precipitate 1. Orange precipitate 1. Brick red precipitate more than 2 g%Due to the fact that an approximate concentration of the reducing sugar can be determined by this test, it is called a semi- quantitative test. Q. 3- What is the alternative test for identification of reducing sugars? Answer- Fehling test is an alternative to Benedict’s test. It differs from Benedict’s test in that it contains sodium potassium Tartrate in place of Sodium citrate and potassium hydroxide as an alkali in place of sodium carbonate in Benedict’s reagent. It is not a preferred test over Benedict’s test since the strong alkali present causes caramalizaton of the sugars; hence it is less sensitive than Benedict’s reagent. Q. 4 – Can Benedict’s test be given positive by non carbohydrate substances? Answer- Any substance which is reducing in nature would give this test positive.  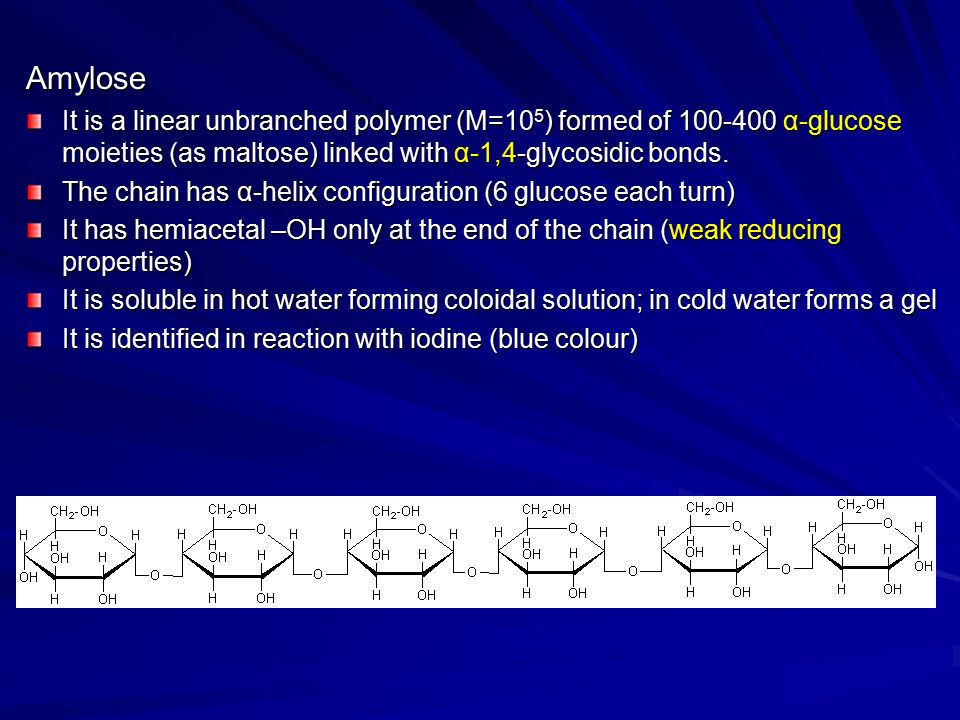
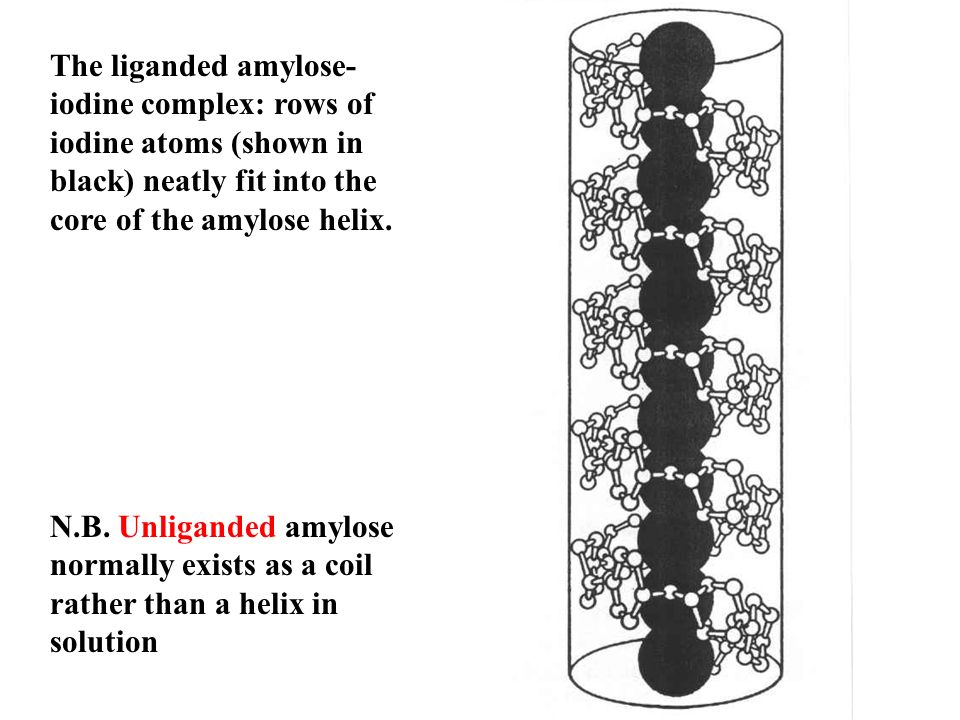
For example- Ascorbates, Urates, Salicylates and Glucuronates give this test as positive, since they are all reducing in nature. Q. 5- Benedict’s and Barfoed’s both are reduction tests, they why are they performed separately ? Answer- Barfoed’s solution contains cupric ions in an acidic medium as compared to Benedict’s test where the medium is alkaline. The milder conditions allow oxidation of mono- saccharides but do not allow oxidation of disaccharides. If the time of heating is carefully controlled, disaccharides do not react while reducing mono- saccharides give the positive result (red Cu. O precipitate). Thus this test is carried out to confirm the presence of mono- saccharides or to rule out the presence of disaccharides. Q. 6- If a sugar gives both Benedicts and Barfoed’s tests negative, which test should be performed to confirm the identity of the sugar? Answer- Both negative tests signify that the sugar is a non reducing disaccharide and hence it could be Sucrose. Seliwanoff and Inversion tests should be carried out to confirm the presence of sucrose. Q. 7- If Benedict test is positive but Barfoed’s test is negative for an unknown sugar solution, which test should be carried out to confirm the identity of the sugar? Answer- Benedict’s positive and Barfoed’s negative shows that the given sugar is reducing in nature but is not a monosaccharide, it could be a disaccharide. Osazone test should be carried out to confirm the identity of the reducing sugar. The characteristic shape of the osazone crystals would signify the identity of the reducing disaccharide. It could be Maltose or Lactose. Maltose forms sun flower shaped crystals while hedge hog shaped crystals are formed by Lactose. Q. 8- An unknown carbohydrate reacted positively with Benedict’s reagent formed a red precipitate with Barfoed’s reagent within 3 minutes and turned blue- green when mixed with Bial’s reagent. What carbohydrate it could be? Answer- From the observations, it is clear that the given carbohydrate is a reducing monosaccharide (Positive Benedict’sand Barfoed’s tests) and since the Bial’s test is a confirmatory test for pentoses so the given carbohydrate could be a pentose. Q. 9- A urine sample gives a positive reaction with Benedict’s and Barfoed’s reagents and forms rhombic plate shaped osazone crystals, what can be the nature of the sugar and what is that clinical state called when the above said sugar is excessively excreted in urine? Answer- Positive reaction with Benedict’s and Barfoed’s reagent signifies that the given carbohydrate is a reducing monosaccharide and rhombic crystals are given by Galactose. Hence it is Galactose and the clinical state is called Galactosuria which is seen in Galactosemia, a congenital disorder of Galactose metabolism. Q. 1. 0- Urine sample of a patient suffering from hereditary fructose intolerance is to be analysed. Fructose is excessively excreted in urine in this condition. Name the conventional tests which should be carried out for its detection? Answer- Fructose is a ketohexose,reducing in nature, hence Seliwanoff test and Benedict’s should be carried out to confirm the presence of fructose. Seliwanoff test is given positive by Keto hexoses in the free or bound form. Q. 1. 1- What is the precaution while performing Barfoed’s test? Answer- This test is used to distinguish reducing monosaccharides from reducing disaccharides by controlling p H and time of heating. Monosaccharides react fast whereas the reaction with disaccharides is slow. However if the heating is prolonged disaccharides can be hydrolysed by acid and the resultant monosaccharides give the test positive. Hence false positive result can be obtained upon prolonged heating of a disaccharide solution. Q. 1. 2- What will be the result of Benedict test with the hydrolytic product of Starch? Answer- The hydrolytic product of starch is maltose and if further hydrolyzed it is Glucose, both are reducing sugars, hence Benedict’s test will be positive in this situation. Q. 1. 3- Sucrose is non reducing in nature but Benedict test shows positive reaction with the hydrolytic products,what is the reason? Answer- Sucrose is a disaccharide having Glucose and Fructose linked together by . Both the functional groups of the components sugars are involved in the linkage, hence no free functional group is there to cause reduction, but upon hydrolysis the products (glucose and fructose ) have free functional groups (Anomeric carbons) to carry out the process of reduction. Hence Benedict’s test is given positive by hydrolytic products. Q. 1. 4- Raffinose is a trisaccharide, what is expected from Barfoed’s test ? Answer- It will be negative; it is given positive only by monosaccharides. Q. 1. 5- Which test should be carried out to confirm the presence of a glycoprotein present in a given solution? Answer- Biuret test for the protein part and Molisch test for the carbohydrate part of the glycoprotein should be carried out for confirmation. Q. 1. 6- Which test out of the followings will be negative with products of hydrolysis of starch Iodine,Benedict, Barfoed’s? Answer- Iodine test will be negative,since iodine forms a coordinate complex between the helically coiled polysaccharide chains and iodine gets centrally located within the helix due to absorption. The color obtained with iodine depends upon the chain length of the polysaccharide (branched or unbranched) required for complex formation. Amylose (Linear chain component) gives a deep blue color; Amylopectin (Branched chain component) gives a purple color, while glycogen gives a reddish- brown color. The hydrolytic products (mono- saccharides) do not react with iodine due to the reasons mentioned above, hence test should be negative. The Structure & Function of Collagen. Shampoo advertisements have made us aware of collagen as a structural protein found in our hair. However, collagen is found in many more tissues. In reality, it makes up approximately 3. The essential role of collagen as a structural component of tissues is made even more apparent by the occurrence of diseases associated with collagen damage. Collagen is found in all the connective and supportive structures of the body. It is found in particularly high amounts in tissues that need to be strong, flexible or both, including cartilage, bones and tendons. Collagen can allow a tissue to withstand immense pulling pressure without stretching - - for example in a tendon transmitting the pull of a muscle to a bone. It can also be elastic, as in skin, and flexible, as in a tendon bending around a joint. Each fiber of collagen contains thousands of individual collagen molecules that are bound together by cross- linking and staggered covalent bonds. Covalent bonds are the strongest bonds that can exist among protein molecules. The collagen molecules themselves are made from 3 individual polypeptides or strings of amino acids. The strands wind around one another in an alpha- helix. The helix forms because of the regular amino acid sequence of the strands. The sequence is a repeating pattern of glycine- proline- X, where X can be any amino acid. Twenty eight different types of collagen have been identified in vertebrates. Collagen types I to IV are the most prevalent. The unique properties of each type are due to segments in the collagen molecules that disrupt the helical structure. These are caused by the amino acids in the X positions of the polypeptide sequence. Different tissues of the body contain different amounts of each type of collagen; for example, cartilage contains a lot of type II, whereas type IV is mostly found in basement membranes. A number of diseases are associated with collagen abnormalities or damage. These can be acquired or caused by a genetic mutation. Ehlers- Danlos syndrome is a genetic disease that results in abnormal collagen production. Symptoms include bruising and hyperelasticity of the skin. Scurvy is an acquired collagen- associated disease caused by vitamin C deficiency. Vitamin C is needed to produce essential amino acids used to make collagen. Without it, collagen fibers cannot be repaired, and blood vessels, tendons and bones become fragile.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2017
Categories |
 RSS Feed
RSS Feed
